Research
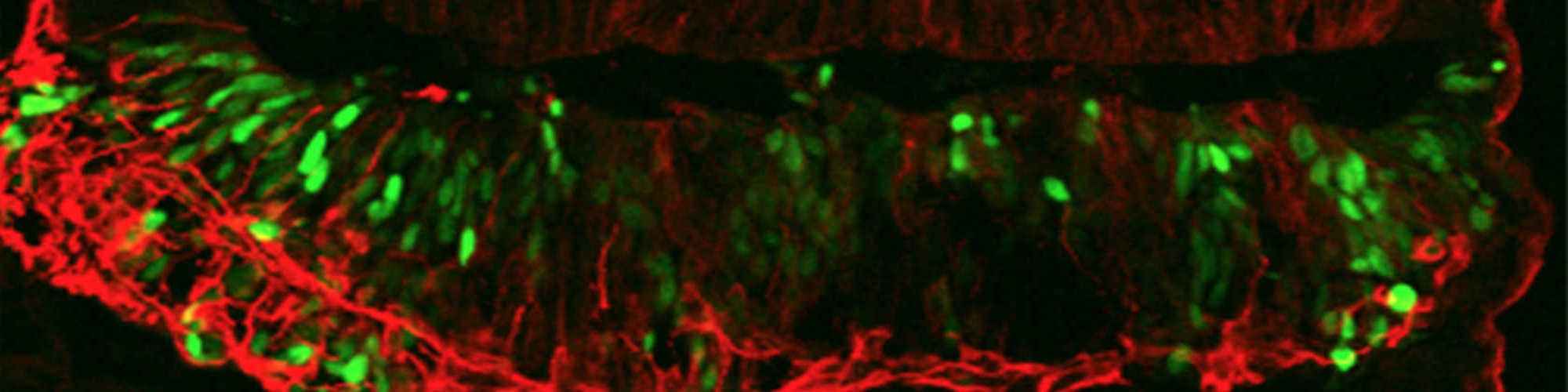
The interest of the lab is to understand and control molecular mechanisms that regulate cell fate. The focus is the evolutionarily conserved Notch pathway, a key regulator of stem cells in most tissues and essential for development. Deregulation of the pathway is the cause of several diseases, most notably cardiovascular diseases, and cancer, where it is linked to aggressive, therapy-resistant cancers.
Key aims of the research group are:
Determine how the microenvironment (chemical, physical and cellular) influences Notch signaling activity and how the integration of cues from the environment and Notch links to cancer progression, stem cell differentiation and response to therapies.
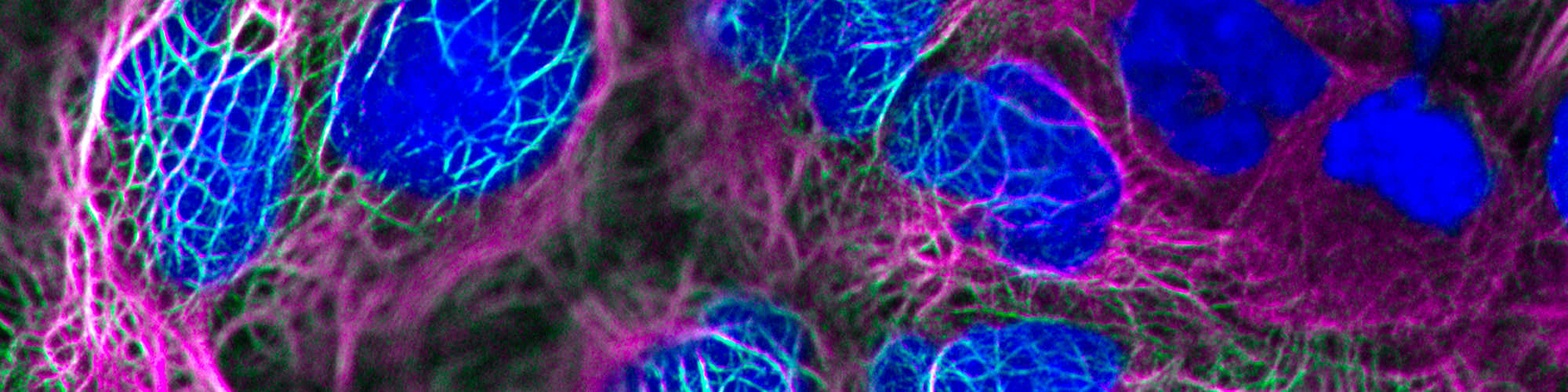
Determine how Notch signaling activity is fine-tuned by spatial control of receptors and ligands, by crosstalk with other signaling pathways and by posttranslational modifications, and how this control translates into Notch-driven cell fate decisions in development, regeneration and disease.
To develop biomaterials for targeted delivery of Notch therapeutics and to develop novel technologies for cell and molecular tracking and control in vitro and in vivo.
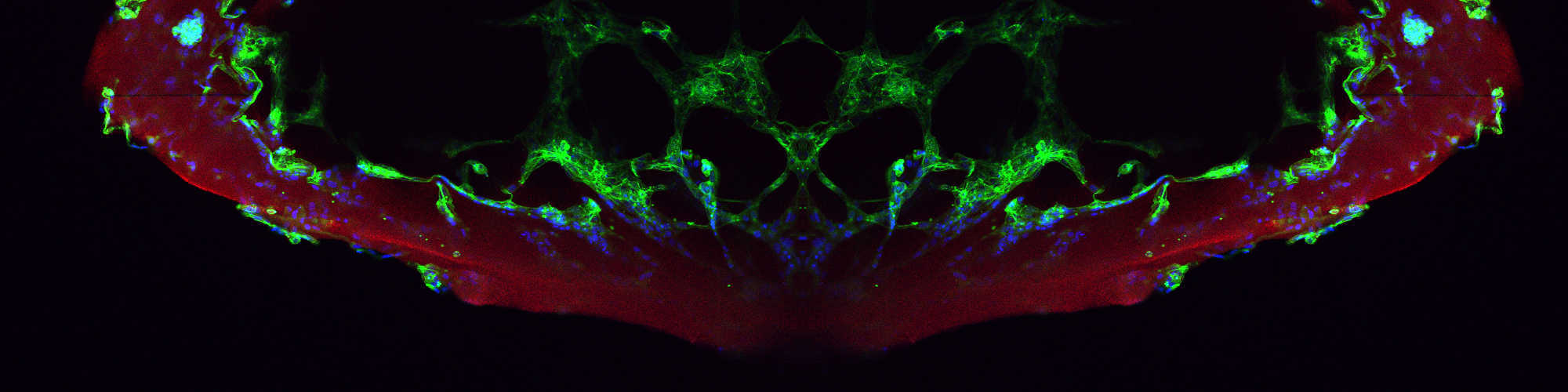
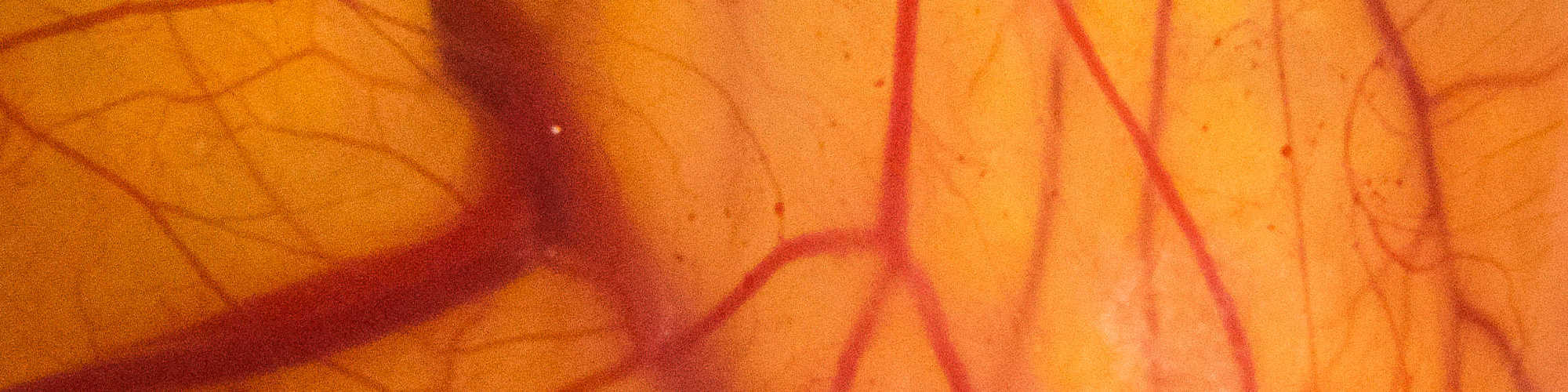
Develop in vivo and biomimetic in vitro model systems for analyses of the role of Notch in cell decisions in tissue homeostasis and disease and for the evaluation of Notch-targeted therapies.
Implement numerical approaches, synthetic technologies and chemical and genetic engineering to enhance our detailed understanding of Notch signaling regulation in development and disease.
Current projects:
Non-animal platform for nanoparticle-based delivery across the blood-brain barrier interface with vehicle evolution, “NAP4DIVE”; Horizon Europe, European Commission
Engineering an integrated platform for generation of human blood stem cells from pluripotent sources, “MakingBlood”; ERC-SyG, European Commission
Tumor and Lymph Node on Chip for cancer studies, Tumor-LN-oC; European Commission (consortium lead by Ioanna Zergioti; ICCS/NTUA, Greece)
Innovation Ecosystem based on the Immune System, InFLAMES; Academy of Finland (consortium lead by Sirpa Jalkanen, University of Turku)
Notch counteracts replication stress to prevent cancer cell senescence; Sigrid Jusélius foundation & Cancer Society of Finland